DiGeorge syndrome results from a chromosomal abnormality. The symptoms and presentation are so diverse and vary so drastically from one child to another that the disease has had a lot of different names in the past, including Shprintzen syndrome, Velocardiofacial syndrome, and CATCH 22 syndrome. DiGeorge syndrome can affect a range of body systems, although presentation varies in severity as well as which body systems are affected. Management depends on these varying factors and can include surgery and therapy.
Cause
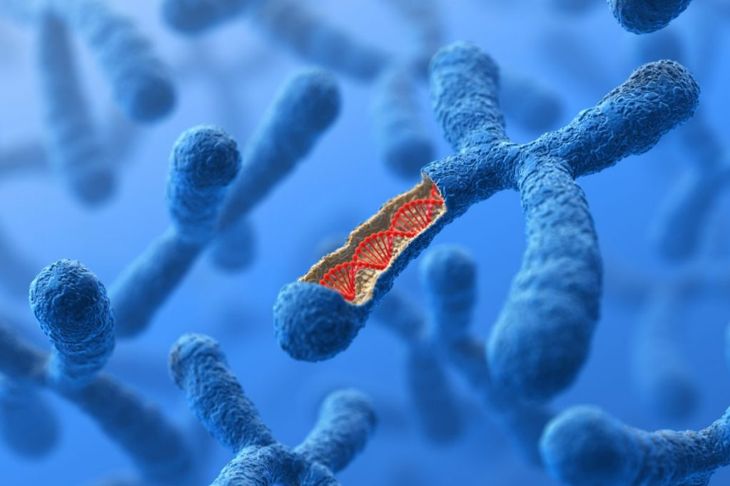
DiGeorge syndrome is a genetic condition caused by a defect in chromosome 22. Specifically, a piece of chromosome 22 is missing in individuals with DiGeorge syndrome. Experts estimate about one in 4,000 people have this genetic abnormality, though some think this number may be even higher because some children present with less severe symptoms.
Genetics
Everyone inherits two copies of chromosome 22, one from each of their parents. DiGeorge syndrome occurs when one of these copies is missing the 11.2 segment, an important piece of the genetic puzzle that includes as many as 40 genes. The deletion occurs randomly and may happen at fertilization or during early fetal development. It is rarely an inherited condition.
Symptoms of DiGeorge Syndrome
Because DiGeorge syndrome can cause as many as 40 genes to be deleted, it affects a lot of body systems. Signs and symptoms can include a heart murmur, frequent infections, a cleft palate, poor muscle tone, and delayed development. A doctor may notice these signs at the child’s birth, or they may become more pronounced throughout infancy and early childhood.
Facial Characteristics
People with DiGeorge syndrome typically present with facial features characteristic of the disease, though some cases are more severe than others. These include small ears that are square in shape, a cleft lip or palate or both, a small mouth and chin, hooded eyelids, and an asymmetric expression when crying.
Heart Defects
Heart defects are a common complication of DiGeorge syndrome, though their specific expression varies. Some children develop a hole between the bottom chambers of the heart (a ventricular septal defect), while others have only one main vessel leading out of the heart instead of two (truncus arteriosus). Infants may also develop a complicated condition involving four separate cardiac defects (tetralogy of Fallot). Signs of these defects include a heart murmur and bluish skin due to low oxygen circulation.
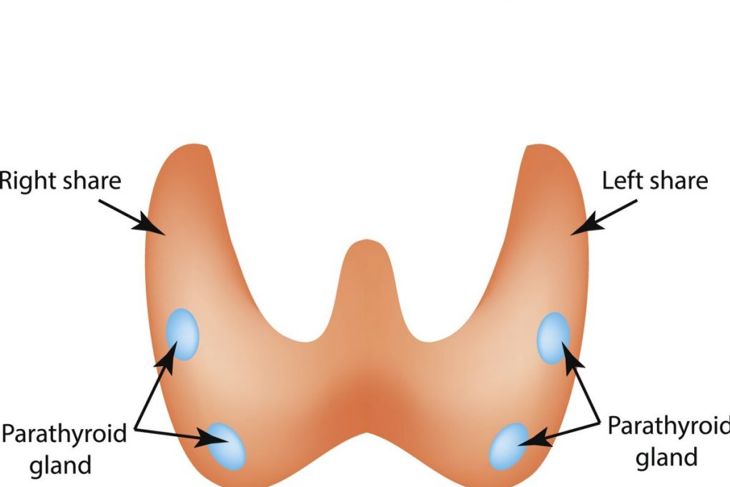
Hypoparathyroidism
The parathyroid glands can be smaller than usual in people with DiGeorge syndrome. These glands are responsible for maintaining calcium levels in the blood. If the condition affects the parathyroid glands, hypocalcemia or low calcium can result, along with elevated phosphorus levels. This often presents as seizures or convulsions. For some people with DiGeorge syndrome, though, parathyroid problems are minor, only becoming a problem during times of high stress. These problems can also decrease in severity over time.
The Thymus
DiGeorge syndrome can also affect the thymus, a gland that stores T-lymphocytes, or T cells, which are part of the immune system responsible for protecting against illness. More specifically, some directly kill viruses while others work with B-lymphocytes to become antibody-producing cells essential for staying healthy. People with DiGeorge syndrome affecting the thymus are at risk for fungal, viral, and bacterial infections and autoimmune diseases. Some may have a small or missing thymus while others may only have a minor deficiency.
Development
DiGeorge syndrome commonly causes significant problems with development, including learning, behavioral, and mental health disorders. Infants and toddlers may not meet developmental milestones on time, including sitting, walking, and talking. Some children with DiGeorge syndrome have an autism spectrum disorder or ADHD. Adults are at a greater risk for anxiety and depression.
Diagnosing DiGeorge Syndrome
Diagnosis is confirmed by a blood test to detect the genetic deletion in chromosome 22. A doctor will order this test if the child shows multiple signs and symptoms of DiGeorge syndrome. Sometimes, a heart defect can be enough to prompt the test, because this is such a common sign of the syndrome. In some cases, the lab may not confirm DiGeorge syndrome despite the indicative symptoms. Treatment will proceed in a similar manner, regardless.
Treatment
There is no cure for DiGeorge syndrome; treatment focuses on managing the symptoms. Interventions commonly include surgery to repair heart defects, calcium and vitamin D supplements to manage hypoparathyroidism, cleft palate repair, and a thymus tissue transplant in the event of severe thymus dysfunction. Physical, occupational, and speech therapy can address developmental delays; problems with feeding, hearing, vision, and growth are managed as they arise.

 Home
Home Health
Health Diet & Nutrition
Diet & Nutrition Living Well
Living Well More
More